Clinical
Clinical Lifecycle Management
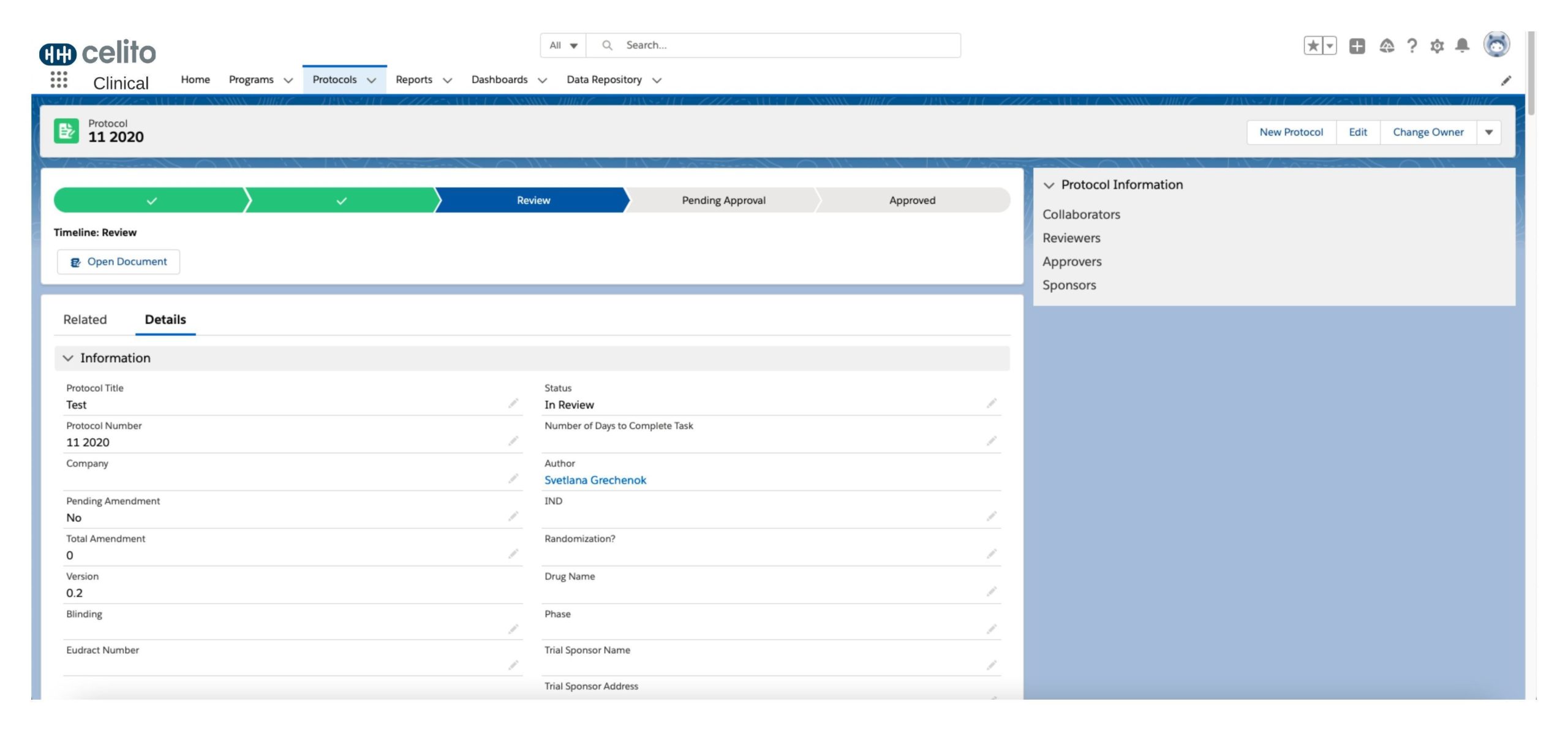
Working through a single pane of glass for your programs and studies brings ease to running your clinical trials. We deliver complete management solutions for digital protocol design, development, and amendments—as well as end-to-end protocol creation and approval workflow. This collaborative authoring works to deliver a single version of truth for your FDA-approval process.
Rather than organizing your pre-clinical data manually, save time and operate with automated and electronic workflows. Modern UI keeps the process intuitive for your team. That way, your pre-clinical work is orchestrated through forms, digital signatures, and data repository.
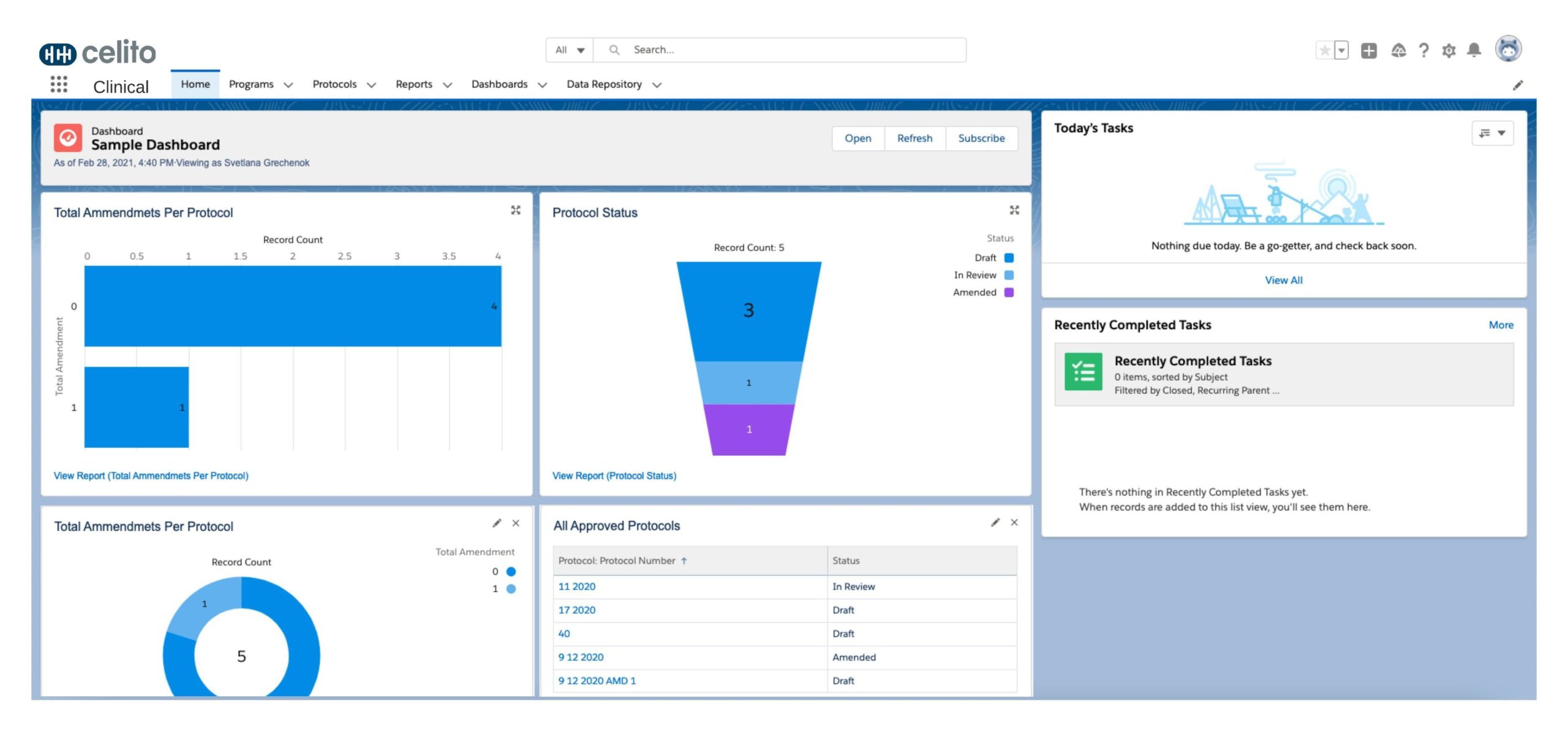
Our clinical life cycle application suite works cross-functionally and streamlines the clinical data exchange between CRO’s and labs. Reviewing operational insights on easy-to-read, dynamic dashboards gives parties the benefit of improved visibility on the state of trials. These measures also help ensure that no information has been corrupted.
A streamlined system works to highlight your product benefits for FDA-approval and a faster move to market. And the right solutions will help deliver well-run and securely-managed clinical data. You only need the proper technology in place.
Studies & Test Design

Protocol Creation & Amendments

IND Preparation

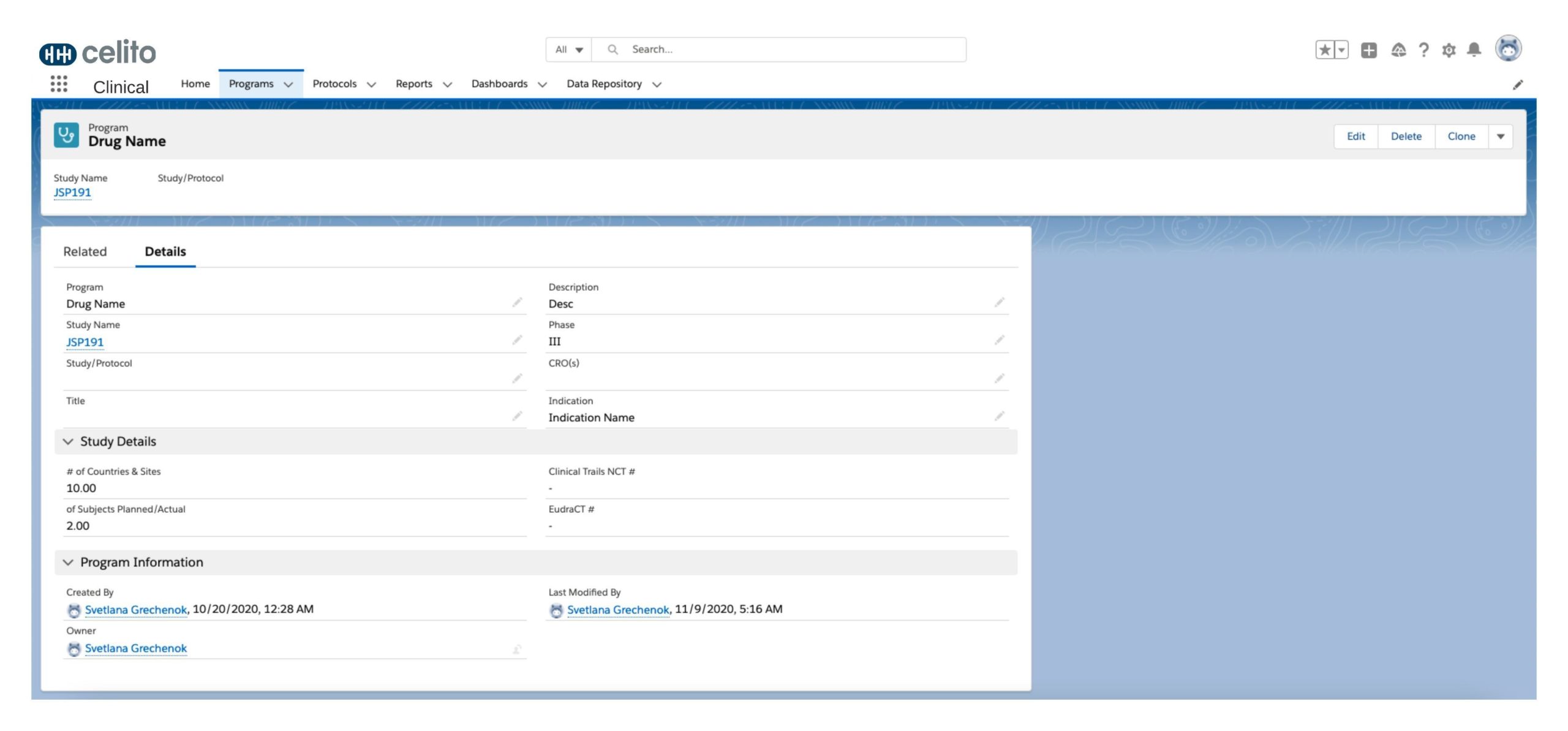
Study Preparation
Digital Study Protocols
Operational Insights

Centralized Clinical Data Hub

Regulatory Doc Repository
We’re here to support your Clinical Process Lifecycle

Celito is a team of experienced IT Executives, Industry Professionals, and Business Consultants focused on the life sciences industry.
Products
Consulting
Company
Celito Tech, Inc.
CORPORATE HEADQUARTERS
2100 Geng Road Suite #210
Palo Alto, CA 94303
CALIFORNIA OFFICE
842 Main St.
Redwood City, CA 94063
+1 650.374.2121
FLORIDA OFFICE
1221 Brickell Avenue Suite #900
Miami, FL 33131
Celito Tech, Inc.
INDIA OFFICE
Celito Tech India Pvt Ltd.
5th Floor,
Regus Grandeur Offices Pvt Ltd,
Caddie Commercial Tower, Aerocity
New Delhi 110037, India
+91 987.011.6939
Privacy Policy | Cookie Policy | Terms of Service | Copyright 2021 © Celito Tech, Inc.